| Catalogue number | C108599 |
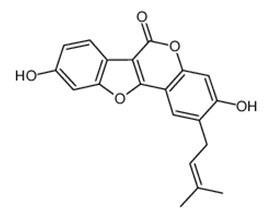
| Chemical name | Psoralidin |
| CAS Number | 18642-23-4 |
| Synonyms | 3,9-dihydroxy-2-(3-methylbut-2-enyl)-6-benzofuro[3,2-c][1]benzopyranone; 3,9-Dihydroxy-2-prenylcoumestan |
| Molecular Weight | C20H16O5 |
| Formula | 336.3 |
| Purity | 98% |
| Physical Description | Powder |
| Solvent | Chloroform, Dichloromethane,DMSO |
| Storage | Stored at 2-8°C, Protected from air and light, refrigerate or freeze |
| Applications | Psoralidin possessing anticancer and chemopreventive properties. Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) triggers apoptosis in cancer cells with no toxicity toward normal tissues. Endogenous TRAIL plays an important role in immune surveillance and defence against cancer cells. Coumarins can modulate TRAIL-mediated apoptosis in cancer cells. We examined the cytotoxic and apoptotic activities of psoralidin in combination with TRAIL on HeLa cancer cells. The cytotoxicity was measured by MTT and LDH assays. The apoptosis was detected using annexin V-FITC staining and mitochondrial membrane potential was evaluated using DePsipher staining by fluorescence microscopy. Death receptor (TRAIL-R1/DR4 and TRAIL-R2/DR5) expression was analyzed using flow cytometry. Psoralidin enhanced TRAIL-induced apoptosis in HeLa cells through increased expression of TRAIL-R2 death receptor and depolarization of mitochondrial membrane potential. Our study indicated that psoralidin augmented the anticancer effects of TRAIL and confirmed a potential use of coumarins in cancer chemoprevention.
|
| References | 1. J. Org. Chem., 2009, 74(7), 2750-2754. 2. Cancer Prev Res (Phila)., 2009, 2(3), 234-243. 3. Molecules, 2012, 17(6), 6449-6464. 4. Progress in neuro-psychopharmacology & biological psychiatry, 2008, 32(2),510-519. |
| Guestbook |
| C262180 | Verbascoside | $72.00/20mg |
| C104147 | Kurarinone | $270.00/10mg |
| C108469 | Tenuifolin | $110.00/20mg |
| C103972 | Xanthohumol | $182.00/10mg |
| C254936 | Quercetin 3-O-β-D Glucoside | $64.00/20mg |
| Size | Price(USD) | Discount |
| 5mg | Inquiry | N/A |
| 10mg | Inquiry | N/A |
| 25mg | Inquiry | N/A |
To place an order, please provide the following information.
1) Your name and telephone number
2) Purchase order number
3) Product number, package size, description, and quantity
4) Shipping and billing addresses
Sent to your order to our email: info@coompo.com

